Menlo Systems product:
Original publication:
Overcoming challenges for THz precision spectroscopy of biomolecules
Researchers from Lille University (France) have used Menlo Systems’ terahertz time-domain spectrometer to investigate biological samples in their novel antenna-waveguide device
The fingerprint region for the detection, identification, and characterization of molecules spans from the infrared (IR) to the terahertz (THz) region of the electromagnetic spectrum, the latter probing collective vibrations of heavy and long molecules. In order to exploit the THz range for spectroscopy of biological samples such as sugars, proteins, DNA, or RNA, scientists search for new solutions which address the specific challenges of a typically tiny sample volume and high water content. A group of researchers from the Institute of Electronics, Microelectronics, and Nanotechnology (IEMN) at Lille University in France presents a novel sensitive technique for broadband precision THz spectroscopy of biological samples making use of field enhancement in a newly designed antenna-waveguide device and Menlo Systems’ terahertz spectrometer.
The working principle of the device is based on increasing the light-matter interaction by confining the electrical field of the THz wave into a volume of a size close to that of the molecules, which are smaller than the THz wavelength (~300 µm) itself. Having the sample positioned directly into the confinement volume increases the coupling efficiency between the THz field and the molecules. The butterfly shaped device consists of a thick slot line waveguide (TSLW) for 2D THz confinement and a specially designed exponentially tapered antenna (ETA) for in- and outcoupling of the THz field.
Besides the size of the waveguide, in the designing of the butterfly device the researchers considered the practicality of its handling. The hollow part must be easily accessible for placement of an analyte, both, solid or liquid, the antenna must allow for injection and extraction of the THz field using standard THz optics, and last, the device should not introduce any significant dispersion or internal reflections.
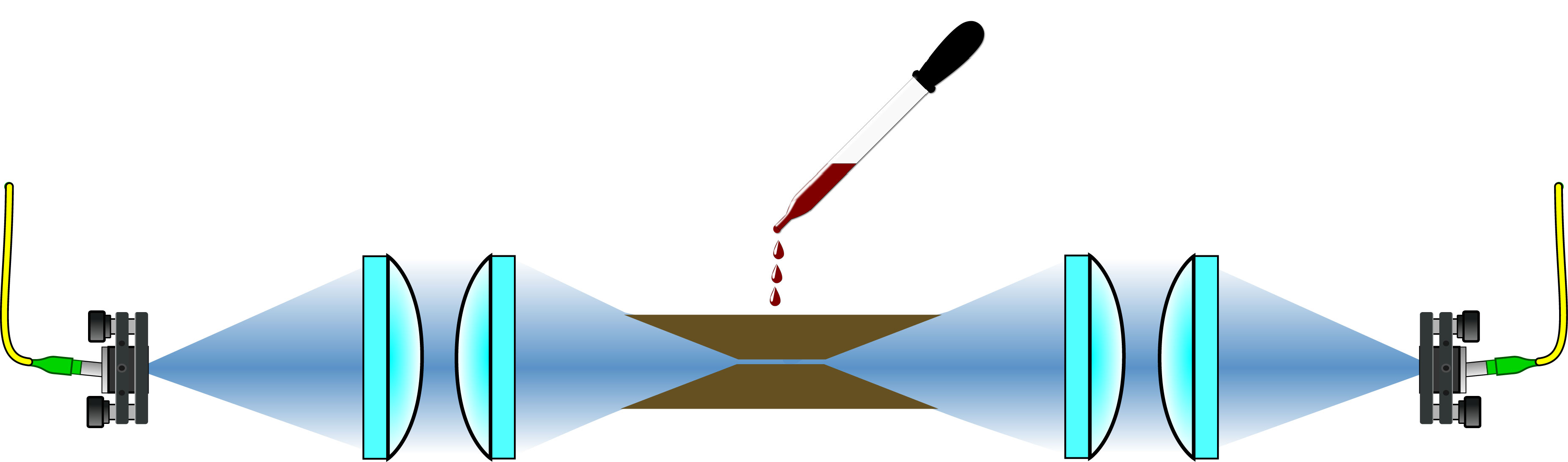
The group performed simulations of the waveguide losses, the antenna far field and reflection characteristics to determine the device’s geometry. This complex pattern served for laser cutting the device from a 500 µm thick copper plate. To achieve vertical and burr-free cutting profiles for undisturbed propagation of the THz light, the device was fabricated by laser ablation. With one side of the butterfly positioned on a micrometer displacement stage, the width of the 10 mm long waveguide is variable.
For characterization, benchmarking and test measurements in dry nitrogen atmosphere the Lille group used Menlo Systems’ TeraSmart Terahertz Time Domain Spectrometer. They found the performance of the butterfly device to meet the requirements for THz time-domain spectroscopy (TDS). Especially in the higher frequency region (above 700 GHz) the losses do not significantly impair the signal quality. A major fraction of the THz field travels through the waveguide, the residual portion can be easily filtered from the THz-TDS data since the waveguide portion is shifted in time due to dispersion.
Lactose, being a common reference substance for THz spectroscopy, served as test substance to verify the functionality and practicality of the butterfly device. First, the sample powder has been inserted into the waveguide to test the use with solid samples. In a second step, a lactose solution was dropped into the waveguide and was left to evaporate. In both cases, the mass of the sample was around 200 µg and below, the volume of the liquid was <10 µl. All measurements reproduce the absorption spectra found in literature. In their work the researchers demonstrate that the new method using the butterfly device in a conventional THz-TDS measurement setup opens the path for the investigation of biological samples, liquid or solid, in minuscule amounts.
Author: Patrizia Krok
Original publication:
R. Peretti et al.: Broadband Terahertz Light–Matter Interaction Enhancement for Precise Spectroscopy of Thin Films and Micro-Samples; Photonics Vol. 5, p. 11 (2018)
