Menlo Systems product:

YLMO Femtosecond Ytterbium Laser
Diagnosis of skin cancer using photon pairs
Sunlight sustains life. However, solar radiation is not always benign, for there are limits to the levels of light that human skin can tolerate. Thus, overexposure to sunlight damages skin cells and can give rise to tumors, as in the case of non-melanoma skin cancer (NMSC, so-called to distinguish this condition from malignant melanoma). A research team led by Prof. Michael G. Giacomelli, Dr. Sherrif Ibrahim, and Vincent D. Ching-Roa in the Department of Biomedical Engineering at the University of Rochester, NY, USA, is currently investigating the efficacy of two-photon fluorescence microscopy as a diagnostic tool for the rapid detection of NMSC. The YLMO laser system supplied by Menlo Systems plays a central role in the project.
Non-melanoma skin cancer is among the most prevalent types of tumor in humans. In fact, in the US, its annual incidence exceeds that of all other malignancies combined. Estimates based on data from the Medicare system indicate that approximately 9600 new cases of NMSC are diagnosed every day. Case numbers of this magnitude underline the need for a rapid, reliable and cost-effective diagnostic test. The current state of the art is based on the histological evaluation of a skin biopsy – and the whole procedure from sample acquisition to diagnosis can take several days or even weeks.
However, thanks to ongoing innovation in medical technology, the advent of novel methods for the identification of skin tumors has begun to change this perspective in recent years. One such advance makes use of confocal laser scanning microscopy, and more recently two-photon fluorescence microscopy (TPFM) has come to the fore.
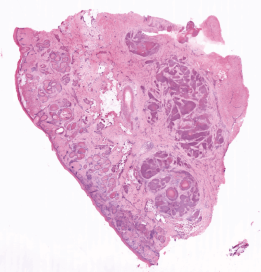
A research group based in the Department of Biomedical Engineering at the University of Rochester, led by Dr. Michael G. Giacomelli and Vincent D. Ching-Roa, has now asked whether TPFM analysis of fresh tissue samples, in combination with real-time imaging, can provide a more efficient option for the diagnosis of non-melanoma skin cancers. If so, patients could receive a rapid and reliable diagnosis of the condition before they leave the doctor’s surgery.
Like confocal laser-scanning microscopy, two-photon fluorescence microscopy is an in-vivo imaging method. “But because it relies on laser light in the near-infrared region, TPFM can penetrate further into the tissue. It can therefore be used for the rapid imaging of freshly obtained, irregularly shaped biopsies following minimal preparation of the sample,” Dr. Giacomelli explains. While confocal laser-scanning microscopy can penetrate to depths of between 20 and 50 µm, multi-photon microscopy can readily visualize tissue at depths of 100 µm or more. This is the major advantage provided by infrared radiation: since less of the incoming light is absorbed or scattered, the laser beam can reach further into the tissue.
Two-photon microscopy captures many of the intrinsic characteristics of biological tissue – including nonlinear refraction index and autofluorescence. In addition, the application of fluorescent stains enables features familiar to pathologists to be specifically labeled, and with the aid of appropriate computer-based analyses, “virtually stained” histological images can be generated that closely resemble those revealed by traditional histological methods. In addition, the non-linear nature of two-photon absorption limits the size of the area of high radiation intensity that is in focus. As a result, the voxels (volume pixels) that make up the image are comparatively small, which in turn enhances the degree of resolution attainable with TPFM.
Giacomelli and his colleagues chose a 1040-nm laser (YLMO) manufactured by Menlo Systems for their initial experiments. The sample is stained with the fluorescent dyes acridine orange and sulforhodamine 101, and scanned by the laser at 16 frames a second. The signals emitted by the two fluorophores (in the 518- to 558-nm and 620- to 680-nm bands, respectively) are then split and filtered prior to detection by two silicon-based photomultipliers. The detectors ensure a high signal-to-noise ratio and are thus compatible with high-speed imaging, as well as robustly reject signals derived from possible contaminants, such as surgical ink. In other work, the team has demonstrated imaging tissue at 50 million pixels per second, also using a YLMO laser.
The results of the study are very encouraging. “We analyzed 15 samples in all. In a high percentage of cases, the diagnosis of non-melanoma skin lesions via two-photon fluorescence microscopy yielded exactly the same results as conventional histological analysis,” says Giacomelli. The diagnoses of basal-cell carcinomas by both methods were 100 % congruent, while cases of squamous cell carcinomas were 89 % accurate.
“Our pilot study suggests that two-photon fluorescence microscopy can identify the characteristic features of non-melanoma skin-cancer lesions that are found in conventional histological samples,” says Vincent D. Ching-Roa. These findings indicate that two-photon fluorescence microscopy has enormous potential for use as a rapid and direct diagnostic alternative to the standard analysis of skin samples. “In addition, the method does not require complex preparation of the sample and dermatologists need no further training in the analysis of results,” Ching-Roa points out.
Nevertheless, as the research team points out, the TPFM-based procedure must be validated in a larger study population in order to accurately assess the diagnostic precision of the approach and its potential impact. “We are now carrying out further clinical studies with the YLMO”, says Giacomelli, “since the technology seems to work as intended, and its cost effectiveness in the clinical context appears to be favorable.”
Author: Thorsten Naeser
Original publications:
V. D. Ching-Roa, Ch. Z. Huang, Sh. F. Ibrahim, B. R. Smoller, M. G. Giacomelli:
Real-time Analysis of Skin Biopsy Specimens With 2-Photon Fluorescence Microscopy;
JAMA Dermatol. 158(10), p. 1175 (2022)
doi:10.1001/jamadermatol.2022.3628
C. Huang, V. D. Ching-Roa, Y. Liu, M. G. Giacomelli:
High-speed mosaic imaging using scanner-synchronized stage position sampling;
J Biomed Opt. 27(1), p. 016502 (2022)
doi: 10.1117/1.JBO.27.1.016502